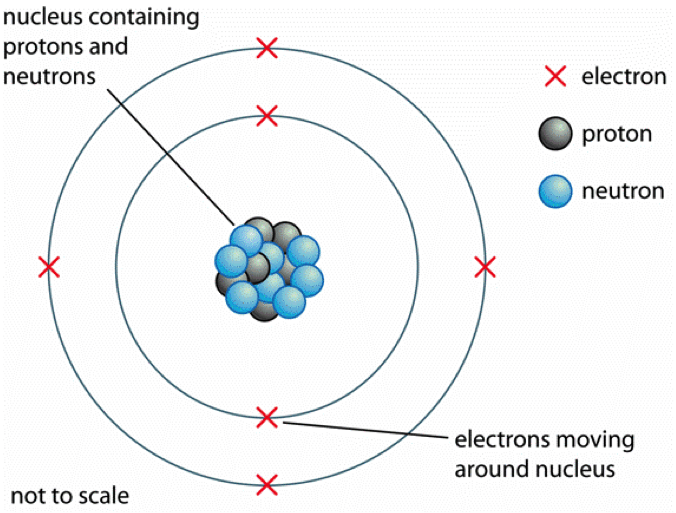
In fact, they have a relative mass of only 1/1840 on the carbon-12 scale. ElectronsĮlectrons are negatively charged particles, and they are much lighter than protons. Neutron numbers can vary between atoms of the same element without much effect on their chemical properties. They also have a relative mass of about 1 and are found alongside protons in the nucleus. Knowing the number of protons in an atom is the key to unlocking its identity! Neutrons The number of protons in an atom is crucial because it determines which element it belongs to on the periodic table. In the centre of an atom, you'll find the nucleus, where protons are tightly packed together. We'll talk about carbon-12 atoms in more detail later on in this article. On this scale, a carbon-12 atom has a mass of exactly 12, and a proton has a mass of approximately 1. Even though they only weigh about kg, their mass is measured on a scale called the carbon-12 scale. Protons are tiny particles that carry a positive charge. So, let's delve deeper into this topic! Protons If you're curious about the world around you, understanding the basics of atomic structure is a great place to start. Atoms are made up of three types of subatomic particles: protons, neutrons, and electrons. 
An atom is the smallest unit of ordinary matter within an element. We now know better! The study of particles inside the atom created the field of atomic structure.Ītomic structure refers to the arrangement of subatomic particles within the atom. However, Democritus believed that atoms were the smallest unit possible. He was partly right! Almost everything around us, from rocks to stars to blood to batteries, is made up of atoms. Democritus, a Greek philosopher, once said that nothing exists except atoms and empty space everything else is just opinion. Atomic structure is a fascinating topic that has been studied for over 2000 years.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
